Health Professional Information
Health Professional Information
How do I use CLEOCIN Vaginal Ovules?
For vaginal use only. Do not take by mouth.
Use one CLEOCIN Vaginal Ovule daily, preferably at bedtime, for 3 days in a row.
Do not use this product if the foiled pouches containing vaginal ovules are torn, opened, or incompletely sealed.
Read the full directions below before using.
Insertion without the applicator:
- 1.
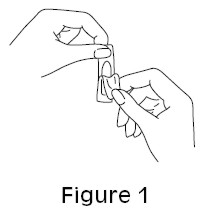
- Remove the vaginal ovule from its packaging (See Figure 1).
- Figure 1
- 2.
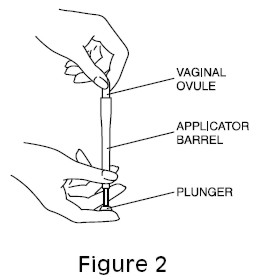
- Holding the ovule with your thumb and a finger, insert it into the vagina (See Figure 2).
Figure 2
- 3.
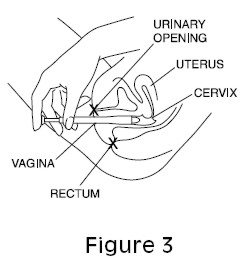
- Using your finger, gently push the ovule into the vagina as far as it will comfortably go (See Figure 3).
Figure 3
- 4.
- Once inside the vagina, the ovule melts. Lie down as soon as possible. This will keep leakage to a minimum.
- 5.
- Repeat steps 1 through 4, before bedtime, for the next 2 days.
STORAGE CONDITIONS:
Store at 25°C (77°F); excursions permitted to 15 – 30°C (59 – 86°F) [see USP Controlled Room Temperature].
Caution: Avoid heat over 30°C (86°F). Avoid high humidity. See end of carton for the lot number and expiration date.
LAB-0649-4.0
Revised February 2026
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.