Health Professional Information
Health Professional Information
DIRECTIONS FOR USE
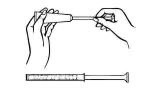
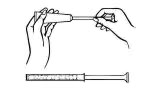
Disposable plastic applicators are provided with this package. They are designed to allow proper vaginal administration of the cream.
Remove cap from cream tube. Screw a plastic applicator on the threaded end of the tube.
Rolling tube from the bottom, squeeze gently and force the medication into the applicator. The applicator is filled when the plunger reaches its predetermined stopping point.
Unscrew the applicator from the tube and replace the cap.
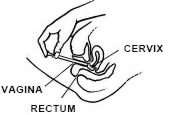
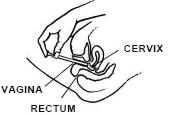
While lying on your back, firmly grasp the applicator barrel and insert into vagina as far as possible without causing discomfort.
Slowly push the plunger until it stops.
Carefully withdraw applicator from vagina, and discard applicator.
REMEMBER TO APPLY ONE APPLICATORFUL EACH NIGHT BEFORE BEDTIME, OR AS PRESCRIBED BY YOUR DOCTOR.
INSTRUCCIONES PARA LA PACIENTE
Este envase contiene aplicadores de plástico desechables. Los aplicadores están diseñados para la administración apropiada de la crema en la vagina.
Remueva la tapa del tubo de crema y enrosque el aplicador de plástico al tubo.
Exprima el tubo suavemente desde el extremo inferior y fuerce el medicamento al aplicador. El aplicador estará lleno cuando el émbolo llega a su máxima longitud.
Desenrosque el aplicador del tubo y vuelva a poner la tapa.
Acuéstese de espalda y agarrando firmemente el aplicador, introdúzcalo en la vagina tanto como sea posible sin causar molestias.
Empuje lentamente el émbolo hasta que se detenga.
Saque el aplicador cuidadosamente de la vagina y descártelo.
RECUERDE APLICARSE UN APLICADOR LLENO TODAS LAS NOCHES AL ACOSTARSE, O DE ACUERDO CON LAS INDICACIONES DE SU MEDICO.
LAB-1058-1.0
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.