Health Professional Information
2 DOSAGE AND ADMINISTRATION
2.1 Dose and Schedule
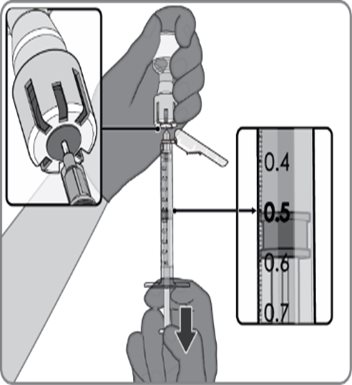
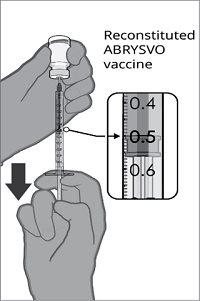
After reconstitution, a single dose of ABRYSVO is either 0.5 mL (Act-O-Vial presentation and vial and vial presentation) or approximately 0.5 mL (vial and prefilled syringe presentation) [see Dosage and Administration (2.2)].
2.2 Presentations and Reconstitution
ABRYSVO is supplied in 3 presentations as follows:
Act-O-Vial Presentation
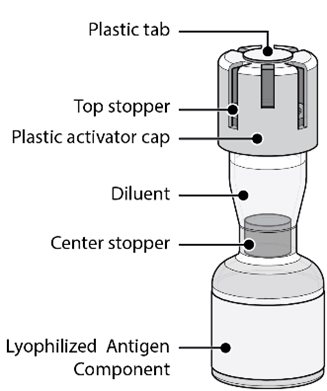
The Act-O-Vial presentation is supplied in cartons. Each Act-O-Vial contains the Lyophilized Antigen Component (a sterile white powder) and Sterile Water Diluent Component.
Vial and Prefilled Syringe Presentation
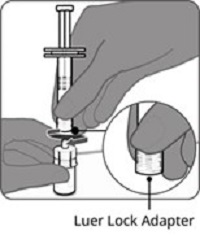
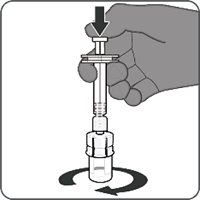
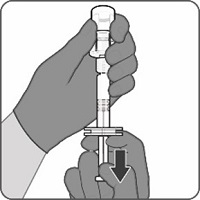
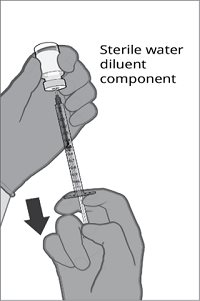
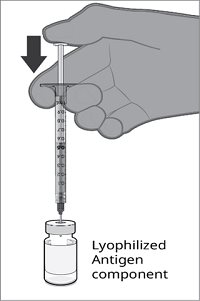
The vial and prefilled syringe presentation is supplied in cartons containing a kit(s). Each kit includes a vial of Lyophilized Antigen Component (a sterile white powder), a prefilled syringe containing Sterile Water Diluent Component, and a vial adapter.
Vial and Vial Presentation
The vial and vial presentation is supplied in cartons that include vials of Lyophilized Antigen Component (a sterile white powder) and vials containing Sterile Water Diluent Component.
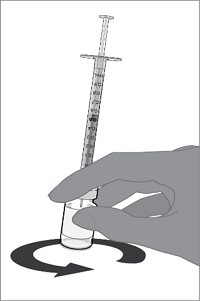
For all presentations, reconstitute the Lyophilized Antigen Component with the Sterile Water Diluent Component to form ABRYSVO, as described in the instructions below.
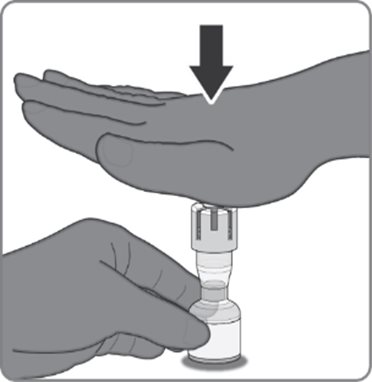
Reconstitution Instructions for the Act-O-Vial Presentation
Reconstitution Instructions for Vial and Prefilled Syringe Presentation
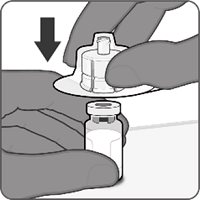
Reconstitution Instructions for the Vial and Vial Presentation
2.3 Administration
For intramuscular use.
After reconstitution, ABRYSVO is a clear and colorless solution. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Discard if either condition is present.
Administer ABRYSVO immediately or store at room temperature [15°C to 30°C (59°F to 86°F)] and use within 4 hours. Discard reconstituted vaccine if not used within 4 hours.
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.