(oxaprozin)
Health Professional Information
Description
11 DESCRIPTION
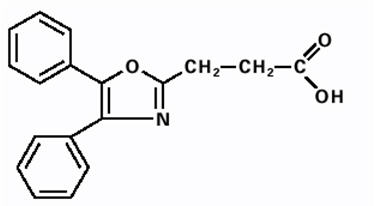
DAYPRO (oxaprozin) caplet is a nonsteroidal anti-inflammatory drug, available as caplets of 600 mg for oral administration. The chemical name is 4,5-diphenyl-2-oxazole-propionic acid. The molecular weight is 293. Its molecular formula is C18H15NO3, and it has the following chemical structure.
Oxaprozin is a white to off-white powder with a slight odor and a melting point of 162°C to 163°C. It is slightly soluble in alcohol and insoluble in water, with an octanol/water partition coefficient of 4.8 at physiologic pH (7.4). The pKa in water is 4.3.
The inactive ingredients in DAYPRO include: microcrystalline cellulose, hypromellose, methylcellulose, magnesium stearate, polacrilin potassium, starch, polyethylene glycol and titanium dioxide. DAYPRO 600-mg caplets are white, capsule-shaped, scored, film-coated, with DAYPRO debossed on one side and 1381 on the other side.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Additional Resources
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.