Health Professional Information
Description
DESCRIPTION
Carboplatin Injection is supplied as a sterile, pyrogen-free, aqueous solution available in 50 mg/5 mL, 150 mg/15 mL, 450 mg/45 mL or 600 mg/60 mL multiple-dose vials containing 10 mg/mL of carboplatin for administration by intravenous infusion. Each mL contains 10 mg carboplatin and Water for Injection, USP.
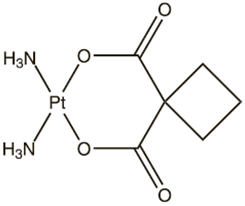
Carboplatin is a platinum coordination compound. The chemical name for carboplatin is platinum, diammine [1,1-cyclobutane-dicarboxylato(2-)-0,0']-,(SP-4-2), and carboplatin has the following structural formula:
Carboplatin is a crystalline powder with the molecular formula of C6H12N204Pt and a molecular weight of 371.25. It is soluble in water at a rate of approximately 14 mg/mL, and the pH of a 1% solution is 5 to 7. It is virtually insoluble in ethanol, acetone, and dimethylacetamide.
Health Professional Information
{{section_name_patient}}
{{section_body_html_patient}}
Additional Resources
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.