Health Professional Information
Description
DESCRIPTION
Acetylcysteine solution is for inhalation (mucolytic agent) or oral administration (acetaminophen antidote), and available as sterile, unpreserved solutions (not for injection).
Acetylcysteine is the N-acetyl derivative of the naturally occurring amino acid, L-cysteine. Chemically, it is N-acetyl-L-cysteine.
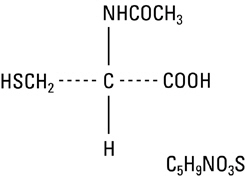
The compound is a white crystalline powder which melts at 104°−110°C and has a very slight odor. The structural formula for acetylcysteine is as follows:
Molecular weight: 163.19
Each mL of the 10% solution contains acetylcysteine 100 mg; edetate disodium, dihydrate 0.25 mg.
Each mL of the 20% solution contains acetylcysteine 200 mg; edetate disodium, dihydrate 0.5 mg.
The solutions also contain sodium hydroxide and may contain hydrochloric acid for pH adjustment, pH 7.0 (6.0 to 7.5). Acetylcysteine Solution, USP is oxygen sensitive.
Acetylcysteine As A Mucolytic Agent |
Additional Resources
Chat online with Pfizer Medical Information regarding your inquiry on a Pfizer medicine or vaccine.
Speak with a Pfizer Medical Information Professional regarding your Pfizer medicine or vaccine inquiry.
Available 9AM-5PM ET Monday to Friday; excluding holidays.
Submit a medical question for a Pfizer medicine or a vaccine.
The submission will be reviewed during our standard business hours.
To report an adverse event related to a Pfizer product and you are not part of a clinical trial* for this medication, click the link below to submit your information:
Pfizer Safety Reporting Site
*If you are involved in a clinical trial for either product, adverse events should be reported to your coordinating study site.
If you cannot use the above website to report an adverse event related to a Pfizer medication, please call (800) 438-1985.
You may also contact the U.S. Food and Drug Administration (FDA) directly to report adverse events or product quality concerns either online at www.fda.gov/medwatch or by calling (800) 332-1088.